原著論文last update : 2026.03.31
2025
1
Kyoya T, Ishida H, Saitoh T, Itoh T*.
Preparation of ethynylsulfonamides and study of their reactivity with nucleophilic amino acids
Org. Biomol. Chem. 2025, 23, 1901−1908. [RSC]
2
Egawa D*, Ishida H, Katakawa K*.
Molecular interactions between piperine and peroxisome proliferator-activated receptor gamma ligand-binding domain revealed using co-crystallization studies.
Acta Cryst. 2025, F81, 201−206. [IUCr]
2024
1
Yano T, Yamada T*, Ishida H, Ohashi N, Itoh T*.
2-cyanopyridine derivatives enable N-terminal cysteine bioconjugation and peptide bond cleavage of glutathione under aqueous and mild conditions
RSC Adv., 2024, 14, 6542-6547. [RSC]
2023
1
Kojima H, Yanagi R, Higuchi E, Yoshizawa M, Shimodaira T, Kumagai M, Kyoya T, Sekine M, Egawa D, Ohashi N, Ishida H, Yamamoto K, Itoh T.
Covalent Modifier Discovery using Hydrogen/Deuterium Exchange–Mass Spectrometry
J. Med. Chem. 2023, 66, 4827–4839. [ACS]
2
Sekine M, Ishida H, Itoh T.
Synthesis of triazolo[c]coumarins and differences in the fluorescence properties of their isomers
Asian J. Org. Chem. 2023, e202300013. [Wiley]
2022
1
Ito T, Miwa C, Haga Y, Kubo M, Itoh T, Yamamoto K, Mise S, Goto E, Tsuzuki T, Matsumura C, Nakano T, Inui H.
Enantioselective metabolism of chiral polychlorinated biphenyl 2, 2′, 3, 4, 4′, 5′, 6-Heptachlorobiphenyl (CB183) by human and rat CYP2B subfamilies
Chemosphere 2022, 308, 136349. [ELSEVIER]
2
Differences in Enantioselective Hydroxylation of 2, 2′, 3, 6-Tetrachlorobiphenyl (CB45) and 2, 2′, 3, 4′, 6-Pentachlorobiphenyl (CB91) by Human and Rat CYP2B Subfamilies
Inui H, Ito T, Miwa C, Haga Y, Kubo M, Itoh T, Yamamoto K, Miyaoka M, Mori T, Tsuzuki H, Mise S, Goto E, Matsumura C, Nakano T
Environ. Sci. & Tec. 2022, 56, 10204-10215.[ACS]
3
Yoshizawa M, Aoyama T, Itoh T, Miyachi H.
Arylalkynyl amide-type peroxisome proliferator-activated receptor γ (PPARγ)-selective antagonists covalently bind to the PPARγ ligand binding domain with a unique binding mode
Bioorg. & Med. Chem. Lett. 2022, 64, 128676. [ELSEVIER]
4
Yabu M, Haga Y, Itoh T, Goto E, Suzuki M, Yamazaki K, Mise S, Yamamoto K, Matsumura C, Nakano T, Sakaki T, Inui H.
Hydroxylation and dechlorination of 3, 3′, 4, 4′-tetrachlorobiphenyl (CB77) by rat and human CYP1A1s and critical roles of amino acids composing their substrate-binding cavity
Sci. Total Environ. 2022, 155848. [ELSEVIER]
5
Ishida H*, Homma S, Kasuga R, Yamamoto K, Itoh T*.
Synthesis of Tetrahydrofuro[2,3-d]oxazoles and Oxazoles by Hypervalent Iodine (III)-Promoted [2 + 2 + 1] Annulation.
Chem. Pharm. Bull. 2022, 70, 192–194. DOI:10.1248/cpb.c21-01001 [CPB]
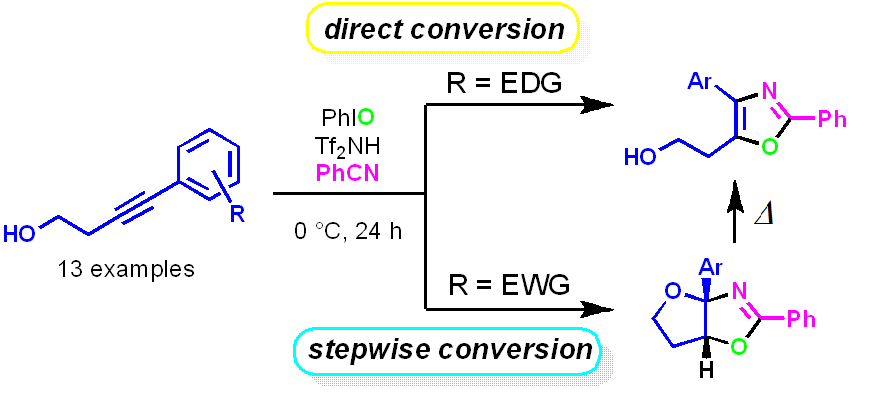
6
Takyo M, Sato Y, Hirata N, Tsuchiya K, Ishida H, Kurohara T, Yanase Y, Ito T, Kanda Y, Yamamoto K, Misawa T*, Demizu Y*.
Oligoarginine-conjugated peptide foldamers inhibiting vitamin D receptor-mediated transcription.
ACS Omega, 2022, 7, 46573–46582. DOI:10.1021/acsomega.2c05409 [ACS]
2021
1
Egawa D, Ogiso T, Nishikata K, Yamamoto K, Itoh T.
Structural Insights into the Loss-of-Function R288H Mutant of Human PPARγ
Biol. Pharm. Bull. 2021, 44, 1196-1201. DOI:10.1248/bpb.b21-00253 [BPB]
2
Yoshikawa C, Ishida H, Ohashi N, Itoh T.
Synthesis of a Coumarin-Based PPARγ Fluorescence Probe for Competitive Binding Assay.
Int. J. Mol. Sci. 2021, 22, 4034. DOI: 10.3390/ijms22084034 [IJMS]
3
Sasaki H, Masuno H, Kawasaki H, Yoshihara A, Numoto, N, Ito N, Ishida H, Yamamoto K, Hirata N, Kanda Y, Kawachi E, Kagechika H, and Tanatani A.
Lithocholic Acid Derivatives as Potent Vitamin D Receptor Agonists.
J. Med. Chem. 2021, 64, 516–526. DOI: 10.1021/acs.jmedchem.0c01420 [ACS]
4
Kanamori S, Ohashi N, Ishida H, Yamamoto K, Itoh T.
HNF4α Is a Covalent Bond-Forming Receptor.
J. Nutr. Sci. Vitaminol., 2021, 67, 126–129. DOI: 10.3177/jnsv.67.126 [JNSV]
5
Yoshikawa C, Ishida H, Ohashi N, Kojima H, Itoh T.
Construction of 7-diethylaminocoumarins promoted by an electro-withdrawing group.
Chem. Pharm. Bull. 2021, 69, 608–611. DOI:10.1248/cpb.c21-00228 [CPB]
Featured Article
2020
1
Montanari R,* Capelli D, Yamamoto K, Awaishima H, Nishikata K, Barendregt A, Heck AJR, Loiodice F, Altieri F, Paiardini A, Grotessi A, Pirone L, Pedone E. M, Peiretti F, Brunel JM, Itoh T,* Pochetti G.*
Insights into PPARγ phosphorylation and its inhibition mechanism.
J. Med. Chem. 2020, 63, 4811–4823. DOI: 10.1021/acs.jmedchem.0c00048 [ACS]
2
Kamata S, Oyama T, Saito K, Honda A, Yamamoto Y, Suda K, Ishikawa R, Itoh T, Watanabe Y, Shibata T, Uchida K, Suematsu M, Ishii I.
PPARα Ligand-Binding Domain Structures with Endogenous Fatty Acids and Fibrates.
iScience. 2020, 23, 101727. DOI:10.1016/j.isci.2020.101727 [Cell]
3
Teratani T, Tomita K, Toma-Fukai S, Nakamura Y, Itoh T, Shimizu H, Shiraishi Y, Sugihara N, Higashiyama M, Shimizu T, Inoue I, Takenaka Y, Hokari R, Adachi T, Shimizu T, Miura S, Kanai T.
Redox-dependent PPARγ/Tnpo1 complex formation enhances PPARγ nuclear localization and signaling.
Free Radic Biol Med. 2020, 156, 45-56. [ELSEVIER]
4
Kojima H,* Itoh T.
Preparation and use of turn-on fluorescent probe for detection and live cell imaging of vitamin D receptor as a target protein.
STAR Protocols, 2020, 1, 100036. DOI: 10.1016/j.xpro.2020.100036 [Cell Press]
5
Yoshikawa C, Ishida H, Itoh T.
Incorporation of a coumarin unit by nucleophilic addition reaction into a PPARγ ligand.
Tetrahedron Lett. 2020, 61, 151842. DOI:10.1016/j.tetlet.2020.151842 [ELSEVIER]
6
Kojima H, Fujita Y, Takeuchi R, Ohashi N, Yamamoto K, Itoh T.
Cyclization Reaction-Based Turn-on Probe for Covalent Labeling of Target Proteins.
Cell Chem. Biol. 2020, 27, 334-349.E11. DOI:10.1016/j.chembiol.2020.01.006 [Cell Press]
Cover Art / Featured Article / Most Read as of 27th Mar 2020
7
Turcotte C, Archambault AS, Dumais É, Martin C, Blanchet MR, Bissonnette E, Ohashi N, Yamamoto K, Itoh T, Laviolette M, Veilleux A, Boulet LP, Marzo VD, Flamand N.
Endocannabinoid hydrolysis inhibition unmasks that unsaturated fatty acids induce a robust biosynthesis of 2-arachidonoylglycerol and its congeners in human myeloid leukocytes.
FASEB J. 2020, 34, 4253-4265. DOI: 10.1096/fj.201902916R [WILEY]
2019
1
Kubo M, Yamamoto K, Itoh T.
Design and synthesis of selective CYP1B1 inhibitor via dearomatization of α-naphthoflavone.
Bioorg. Med. Chem. 2019, 27, 285–304. doi.org/10.1016/j.bmc.2018.11.045. [BMC]
2018
1
Kanamori S, Ishida H, Yamamoto K, Itoh T.
Construction of a series of intermediates in the β-oxidation pathway from THA to EPA via DHA in free acid form.
Bioorg. Med. Chem. 2018, 26, 4390–4401. doi:10.1016/j.bmc.2018.07.004. [BMC]
2
Goto E, Haga Y, Kubo M, Itoh T, Kasai C, Shoji O, Yamamoto K, Matsumura C, Nakano T, Inui H.
Metabolic enhancement of 2,3´,4,4´,5-pentachlorobiphenyl (CB118) using cytochrome P450 monooxygenase isolated from soil bacterium under the presence of perfluorocarboxylic acids (PFCAs) and the structural basis of its metabolism.
Chemosphere, 2018, 210, 376–383. doi:10.1016/j.chemosphere.2018.07.026. [ELSEVIER]
3
Yoshizawa M, Itoh T, Hori T, Kato A, Anami Y, Yoshimoto N, Yamamoto K.
Identification of the histidine residue in vitamin D receptor that covalently binds to electrophilic ligands.
J. Med. Chem. 2018, 61, 6339–6349. doi:10.1021/acs.jmedchem.8b00774. [ACS]
4
Itoh T,* Yoshimoto N, Hirano Y, Yamamoto K.*
Structural basis for the selective inhibition of activated thrombin-activatable fibrinolysis inhibitor (TAFIa) by a selenium-containing inhibitor with chloro-aminopyridine as a basic group.
Bioorg. Med. Chem. Lett. 2018, 28, 2256–2260. doi: 10.1016/j.bmcl.2018.05.042. [BMCL]
Cover Art
2017
1
Kojima H, Itoh T, Yamamoto K.
On-site reaction for PPARγ modification using a specific bifunctional ligand.
Bioorg. Med. Chem. 2017, 25, 6492–6500. DOI: 10.1016/j.bmc.2017.10.024. [BMC]
2
Kato A, Yamao M, Hashihara Y, Ishida H, Itoh T, Yamamoto K.
Vitamin D analogues with a p-hydroxyphenyl group at the C25 position: crystal structure of vitamin D receptor ligand-binding domain complexed with the ligand explains the mechanism underlying full antagonistic action.
J. Med. Chem. 2017, 60, 8394–8406. DOI: 10.1021/acs.jmedchem.7b00819. [ACS].
3
Yamamoto Y, Itoh T, Yamamoto K.
A study of synthetic approaches to 2-acyl DHA lysophosphatidic acid.
Org. Biomol. Chem. 2017, 15, 8186–8192. DOI: 10.1039/c7ob01771e. [RSC]
4
Ohura A, Itoh T, Ishida H, Saito A, Yamamoto K.
Three-Component Regioselective Synthesis of Tetrahydrofuro[2,3-d]oxazoles and their Efficient Conversion to Oxazoles.
Asian J. Org. Chem. 2017, 6, 673–676. DOI: 10.1002/ajoc.201700074 [ACES]
5
Egawa D, Itoh T, Kato A, Kataoka S, Anami Y, Yamamoto K.
SRC2-3 binds to vitamin D receptor with high sensitivity and strong affinity.
Bioorg. Med. Chem. 2017, 25, 568–574. DOI: 10.1016/j.bmc.2016.11.020 [BMC]
2016
1
Anami Y, Shimizu N, Ekimoto T, Egawa D, Itoh T, Ikeguchi M, Yamamoto K.
Apo- and Antagonist-Binding Structures of Vitamin D Receptor Ligand-Binding Domain Revealed by Hybrid Approach Combining Small-Angle X‑ray Scattering and Molecular Dynamics.
J. Med. Chem. 2016, 59, 7888−7900. DOI: 10.1021/acs.jmedchem.6b00682. [ACS]
2
Egawa D, Itoh T, Akiyama Y, Saito T, Yamamoto K.
17-oxoDHA is a PPARα/γ dual covalent modifier and agonist.
ACS Chem. Biol. 2016, 11, 2447−2455. DOI: 10.1021/acschembio.6b00338 [ACS]
3
Kato A, Anami Y, Egawa D, Itoh T, Yamamoto K.
Helix12-stabilization antagonist of vitamin D receptor.
Bioconjugate Chem. 2016, 27, 1750−1761. DOI: 10.1021/acs.bioconjchem.6b00246[ACS]
4
Mise S, Haga Y, Itoh T, Kato A, Fukuda I, Goto E, Yamamoto K, Yabu M, Matsumura C, Nakano T, Sakaki T, Inui H.
Structural determinants of the position of 2,3’,4,4’,5-pentachlorobiphenyl (CB118) hydroxylation by mammalian cytochrome P450 monooxygenases.
Toxicol. Sci. 2016, 152, 340−348. doi: 10.1093/toxsci/kfw086 [PubMed]
5
Itoh T, Saito T, Yamamoto Y, Ishida H, Yamamoto K.
Gram scale synthesis of specialized pro-resolving mediator 17(S)-HDHA using lipoxygenase enhanced by water-soluble reducing agent TCEP.
Bioorg. Med. Chem. Lett. 2016, 26, 343-345. Doi:10.1016/j.bmcl.2015.12.011. [BMCL]
2015
1
Anami Y, Sakamaki Y, Itoh T, Inaba Y, Nakabayashi M, Ikura T, Ito N, Yamamoto K.
Fine tuning of agonistic/antagonistic activity for vitamin D receptor by 22-alkyl chain length of ligands: 22S-Hexyl compound unexpectedly restored agonistic activity.
Bioorganic. Med. Chem. 2015, 23, 7274–81. doi:10.1016/j.bmc.2015.10.026. [BMC]
2
Yamamoto Y, Itoh T, Yamamoto K.
Chemical Synthesis of a Very Long Chain Fatty Acid, Hexacosanoic Acid (C26:0), and the Ceramide Containing Hexacosanoic Acid.
J. Nutr. Sci. Vitaminol. 2015, 61, 222–227. doi: 10.3177/jnsv.61.222. [JNSV]
3
Egawa D, Itoh T, Yamamoto K.
Characterization of covalent bond formation between PPARγ and oxo-fatty acids.
Bioconjugate Chem. 2015, 26, 690-698. DOI: 10.1021/acs.bioconjchem.5b00021. [ACS]
2014
1
Anami Y, Itoh T, Egawa D, Yoshimoto N, Yamamoto K.
A mixed population of antagonist and agonist binding conformers in a single crystal explains partial agonism against vitamin D receptor: Active vitamin D analogues with 22R-alkyl group.
J. Med. Chem. 2014, 57, 4351-4367. DOI: 10.1021/jm500392t [ACS]
2
Rhieu SY, Annalora AJ, Laporta E, Welsh J, Itoh T, Yamamoto K, Sakaki T, Chen TC, Uskokovic MR, Reddy GS.
Potent antiproliferative effects of 25-hydroxy-16-ene-23-yne-vitamin D3 that resists the catalytic activity of both CYP27B1 and CYP24A1.
J. Cell. Biochem. 2014, 115, 1392-1402. doi: 10.1002/jcb.24789. [JCB]
2013
1
Yoshimoto N, Itoh T, Inaba Y, Ishii H, Yamamoto K.
Structural basis for inhibition of carboxypeptidase B by selenium-containing inhibitor: Selenium coordinates to zinc in enzyme.
J. Med. Chem. 2013, 56, 7527-7535. [ACS]
2012
1
Yoshimoto N, Sasaki T, Sugimoto K, Ishii H, Yamamoto K.
Design and characterization of a selenium-containing inhibitor of activated thrombin-activatable fibrinolysis inhibitor (TAFIa), a zinc-containing metalloprotease.
J. Med. Chem. 2012, 55, 7696-7705. [ACS] [PubMed]
2
Sasaki T, Yoshimoto N, Sugimoto K, Takada K, Murayama N, Yamazaki H, Yamamoto K, Ishii H.
Intravenous and oral administrations of DD2 [7-Amino-2-(sulfanylmethyl)heptanoic acid] produce thrombolysis through inhibition of plasma TAFIa in rats with tissue factor-induced microthrombosis.
Thromb Res. 2012, 130, e222-e228. [PubMed]
3
Yasuda K, Ikushiro S, Wakayama S, Itoh T, Yamamoto K, Kamakura M, Munetsuna E, Ohta M, Sakaki T.
Comparison of Metabolism of Sesamin and Episesamin by Drug Metabolizing Enzymes in Human Liver.
Drug Metab Dispos, 2012, 40, 1917-1926. [PubMed]
4
Yoshimoto N, Sakamaki Y, Haeta M, Kato A, Inaba Y, Itoh T, Nakabayashi M, Ito N, Yamamoto K.
Butyl pocket formation in the vitamin D receptor strongly affects the agonistic or antagonistic behavior of ligand.
J. Med. Chem. 2012, 55, 4373-4381. [ACS] [PubMed]
2011
1
Iglesias-Gato D, Zheng S, Flanagan JN, Jiang L, Kittaka A, Sakaki T, Yamamoto K, Itoh T, LeBrasseur NL,Norstedt G, Chen TC.
Substitution at carbon 2 of 19-nor-1α,25-dihydroxyvitamin D3 with 3-hydroxypropyl group generates an analog with enhanced chemotherapeutic potency in PC-3 prostate cancer cells.
J. Steroid Biochem. Mol. Biol. 2011, 127, 269-275.[PubMed]
2
Itoh T, Tomiyasu A, Yamamoto K.
Efficient Synthesis of the Very-Long-Chain n-3 Fatty Acids, Tetracosahexaenoic acid (C24:6n-3) and Tricosahexaenoic acid (C23:6n-3).
Lipids. 2011, 46, 455-461. [PubMed]
3
Yamazaki K, Suzuki M, Itoh T, Yamamoto K, Kanemitsu M, Matsumura C, Nakano T, Sakaki T, Fukami Y, Imaishi H, Inui H.
Structural basis of species differences between human and experimental animal CYP1A1s in metabolism of 3,3’,4,4’,5-pentachlorobiphenyl.
J. Biochem. 2011, 149, 487-494. [PubMed]
2010
1
Sakamaki Y, Inaba Y, Yoshimoto N, Yamamoto K.
Potent Antagonist for the Vitamin D Receptor: Vitamin D Analogues with Simple Side Chain Structure.
J. Med. Chem. 2010, 53, 5813-5826. [ACS]
2
Takemura H, Itoh T, Yamamoto K, Sakakibara H, Shimoi K.
Selective inhibition of methoxyflavonoids on human CYP1B1 activity.
Bioorg. Med. Chem. 2010, 18, 6310-6315. [PubMed]
3
Itoh T, Takemura H, Shimoi K, Yamamoto K.
A 3D model of CYP1B1 explains the dominant 4-hydroxylation of estradiol.
J. Chem. Inf. Model. 2010, 50, 1173-1178. [ACS]
4
Yamagishi K, Yamamoto K, Mochizuki Y, Nakano T, Yamada S, Tokiwa H.
Flexible Ligand Recognition of Peroxisome Proliferator-Activated Receptor-γ (PPARγ).
Bioorg. Med. Chem. Lett. 2010, 20, 3344-3347. [PubMed]
5
Inaba Y, Nakabayashi M, Itoh T, Yoshimoto N, Ikura T, Ito N, Shimizu M, Yamamoto K.
22S-Butyl-1α,24R-dihydroxyvitamin D3: Recovery of vitamin D receptor agonistic activity.
J. Steroid Biochem. Mol. Biol. 2010, 121, 146-150. [PubMed]
6
Itoh T, Yoshimoto N, Yamamoto K.
Synthesis of oxidized fatty acid derivatives via an iodolactonization reaction.
Heterocycles. 2010, 80, 689-695. [link]
2009
1
Tsai N-P, Huq M, Gupta P, Yamamoto K, Kagechika H, Wei L-N.
Activation of Testicular Orphan Receptor 4 by Fatty Acids.
Biochim. Biophys. Acta, Gene Regul. Mech. 2009, 1789, 734-740. [PubMed]
2
Yoshida Y, Mohri K, Isobe K, Itoh T, Yamamoto K.
Biomimetic Total Synthesis of (±)-8-Oxoerymelanthine.
J. Org. Chem. 2009, 74, 6010-6015. [ACS]
3
Inaba Y, Yoshimoto N, Sakamaki Y, Nakabayashi M, Ikura T, Tamamura H, Ito N, Shimizu M, Yamamoto K.
A New Class of Vitamin D Analogues that Induce Structural Rearrangement of the Ligand-Binding Pocket of the Receptor.
J. Med. Chem. 2009, 52, 1438–1449. [ACS]
2008
1
Itoh T, Fairall L, Amin K, Inaba Y, Szanto A, Balint BL, Nagy L, Yamamoto K, Schwabe J. W. R.
Structural basis for the activation of PPARγ by oxidized fatty acids.
Nat. Struct. Mol. Biol. 2008, 15, 924-931. [Nature]
2
Yamamoto K, Ninomiya Y, Iseki M, Nakachi Y, Kanesaki-Yatsuka Y, Yamanoue Y, Itoh T, Nishii Y, PetrovskyN, Okazaki Y.
4-Hydroxydocosahexaenoic acid, a potent peroxisome proliferator-activated receptor g agonist alleviates the symptoms of DSS-induced colitis.
Biochem. Biophys. Res. Commun. 2008, 367, 566-572. [PubMed]
3
Takaku H, Miyamoto Y, Asami S, Shimazaki M, Yamada S, Yamamoto K, Udagawa N, DeLuca HF, Shimizu M.
Synthesis and structure-activity relationships of 16-ene-22-thia-1α,25-dihydroxy-26,27-dimethyl-19-norvitamin D3 analogs having side chains of different sizes.
Bioorg. Med. Chem. 2008, 16, 1796-1815. [PubMed]
4
Yoshimoto N, Inaba Y, Yamada S, Makishima M, Shimizu M, Yamamoto K.
2-Methylene 19-nor-25-dehydro-1α-hydroxyvitamin D3 26,23-lactones: Synthesis, biological activities and molecular basis of passive antagonism.
Bioorg. Med. Chem. 2008, 16, 457-473. [PubMed]
2007
1
Urushino N, Nakabayashi S, Arai M, Kittaka A, Chen TC, Yamamoto K, Hayashi K, Kato S, Ohta M, Kamakura M, Ikushiro S, Sakaki T.
Kinetic Studies of 25-Hydroxy-19-Nor-Vitamin D3 and 1α,25-Dihydroxy-19-Nor-Vitamin D3 Hydroxylation by CYP27B1 and CYP24A1.
Drug Metab. Dispos. 2007, 35, 1482-1488. [PubMed]
2
Inaba Y, Yamamoto K, Yoshimoto N, Matsunawa M, Uno S, Yamada S, Makishima M.
Vitamin D3 derivatives with adamantane or lactone ring side chains are cell type-selective vitamin D receptor modulators.
Mol. Pharmacol. 2007, 71, 1298-1311. [PubMed]
3
Yamamoto K, Inaba Y, Yoshimoto N, Choi M, DeLuca HF, Yamada S.
22-Alkyl-20-epi-1α,25-dihydroxyvitamin D3 Compounds of Super-agonistic Activity: Syntheses, Biological Activities and Interaction with the Receptor.
J. Med. Chem. 2007, 50, 932-939. [ACS]
4
Sawada N, Yamamoto K, Yamada S, Ikushiro S, Kamakura M, Ohta M, Inouye K, Sakaki T.
Unique enzymatic properties of the mutant Q85H of human CYP27A1 in vitamin D hydroxylation: Gln 85 is responsible for both substrate-binding and protein folding.
Biochem. Biophys. Res. Commun., 2007, 355, 211-216. [link]
5
Igarashi M, Yoshimoto N, Yamamoto K, Shimizu M, Makishima M, DeLuca HF, Yamada S.
Identification of a highly potent vitamin D receptor antagonist: (25S)-26-Adamantyl-25-hydroxy-2-methylene-22,23-didehydro-19,27-dinor-20-epi-vitamin D3 (ADMI3).
Arch. Biochem. Biophys. 2007, 460, 240-253. [PubMed]
6
Yamamoto K, Choi M, Abe D, Shimizu M, Yamada M.
Alanine scanning mutational analysis of the ligand binding pocket of the human vitamin D receptor.
J. Steroid Biochem. Mol. Biol. 2007, 103, 282-285. [PubMed]
7
Kobayashi E, Shimazaki M, Miyamoto Y, Masuno H, Yamamoto K, DeLuca HF, Yamada S, Shimizu M.
Structure-activity relationships of 19-norvitamin D analogs having a fluoroethylidene group at the C-2 position.
Bioorg. Med. Chem. 2007, 15, 1475-1482. [PubMed]